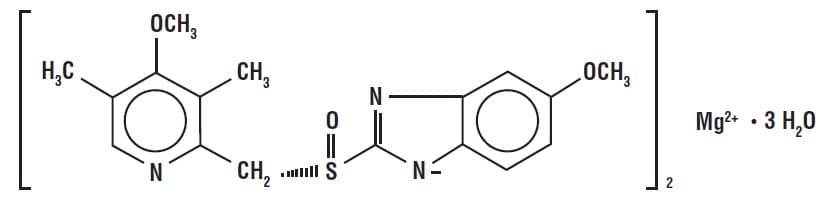
Naproxen and esomeprazole magnesium
20 mg/1 / 500 mg/1 · Delayed-Release Tablet
By PHOENIX RX LLC
- NDC
- 85509-1438
- Route
- Oral
- Therapeutic class
- Anti-Inflammatory Agents, Non-Steroidal [CS], Cyclooxygenase Inhibitors [MoA], Cytochrome P450 2C19 Inhibitors [MoA], Nonsteroidal Anti-inflammatory Drug , Proton Pump Inhibitor , Proton Pump Inhibitors [MoA]
- Marketing category
- ANDA
Package sizes
- 30 TABLET, DELAYED RELEASE in 1 BOTTLE (85509-1438-3)NDC 85509-1438-3Available to order
- 60 TABLET, DELAYED RELEASE in 1 BOTTLE (85509-1438-6)NDC 85509-1438-6Available to order
- 90 TABLET, DELAYED RELEASE in 1 BOTTLE (85509-1438-9)NDC 85509-1438-9Available to order
- 100 TABLET, DELAYED RELEASE in 1 BOTTLE (85509-1438-1)NDC 85509-1438-1Available to order
Additional information
Full prescribing information for this product is available on request. Submit a sourcing request and our team will share lot details, dating, and current pricing.
Request this medication
Send a request and we'll respond with availability, lot details, and pricing within 2 business hours.
Related in Analgesics & Pain Relief
See all →RAPID RELIEF (0.025% Capsaicin Pain Patch)
.00025 g/g · Patch
83569
Available to source
10% Methyl Salicylate Pain Relief Patch (10% Methyl Salicylate)
10 mg/100mg · Tape
Akron Coating & Adhesives
Available to source

Extra Strength Nite-Time Pain Relief
500 mg/1 / 25 mg/1 · Tablet
WALGREENS
Available to source
Abdominal Pain Drops
Liquid
Professional Complementary Health Formulas
Available to source
