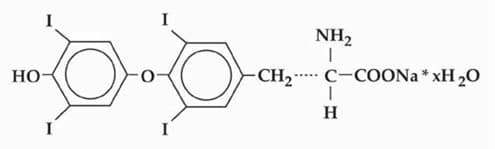
Raloxifene Hydrochloride
60 mg/1 · Film-Coated Tablet
By Aurobindo Pharma Limited
- NDC
- 65862-709
- Route
- Oral
- Therapeutic class
- Estrogen Agonist/Antagonist , Selective Estrogen Receptor Modulators [MoA]
- Marketing category
- ANDA
Package sizes
- 3 BLISTER PACK in 1 CARTON (65862-709-03) / 10 TABLET, FILM COATED in 1 BLISTER PACK (65862-709-10)NDC 65862-709-03Available to order
- 30 TABLET, FILM COATED in 1 BOTTLE (65862-709-30)NDC 65862-709-30Available to order
- 100 TABLET, FILM COATED in 1 BOTTLE (65862-709-01)NDC 65862-709-01Available to order
- 1000 TABLET, FILM COATED in 1 BOTTLE (65862-709-99)NDC 65862-709-99Available to order
- 2000 TABLET, FILM COATED in 1 BOTTLE (65862-709-22)NDC 65862-709-22Available to order
Additional information
Full prescribing information for this product is available on request. Submit a sourcing request and our team will share lot details, dating, and current pricing.
Request this medication
Send a request and we'll respond with availability, lot details, and pricing within 2 business hours.
Related in Diabetes & Endocrine
See all →
OMEOSPORT
Pellet
Guna spa
Available to source
GUNA-GERIATRICS
Solution / Drops
Guna spa
Available to source
PROCORT (1.85% hydrocortisone acetate - 1.15% pramoxine hci cream)
18.5 mg/g / 11.5 mg/g · Cream
Womens Choice Pharmaceuticals LLC
Available to source

Equate Hydrocortione (1% Hydrocortisone)
10 mg/g · Cream
Walmart Inc.
Available to source
