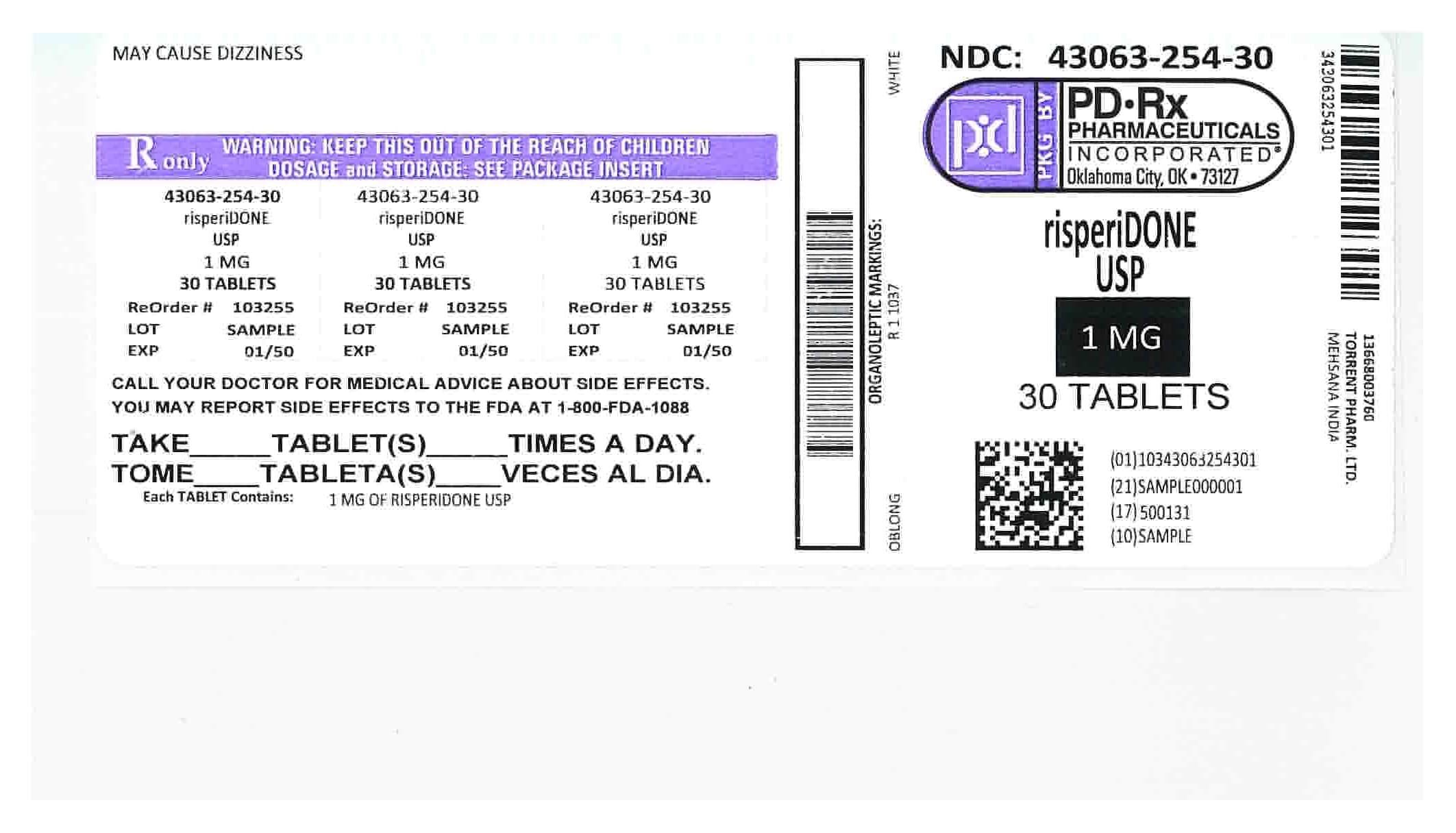
Risperidone
3 mg/1 · Tablet Orally Disintegrating
By Sun Pharmaceutical Industries, Inc.
- NDC
- 63304-643
- Route
- Oral
- Therapeutic class
- Atypical Antipsychotic
- Marketing category
- ANDA
Package sizes
- 1 BLISTER PACK in 1 CARTON (63304-643-69) / 10 TABLET, ORALLY DISINTEGRATING in 1 BLISTER PACKNDC 63304-643-69Available to order
- 10 POUCH in 1 BOX, UNIT-DOSE (63304-643-17) / 1 TABLET, ORALLY DISINTEGRATING in 1 POUCH (63304-643-11)NDC 63304-643-17Available to order
- 30 TABLET, ORALLY DISINTEGRATING in 1 BOTTLE (63304-643-30)NDC 63304-643-30Available to order
- 500 TABLET, ORALLY DISINTEGRATING in 1 BOTTLE (63304-643-05)NDC 63304-643-05Available to order
Additional information
Full prescribing information for this product is available on request. Submit a sourcing request and our team will share lot details, dating, and current pricing.
Request this medication
Send a request and we'll respond with availability, lot details, and pricing within 2 business hours.
Related in Ophthalmic & Otic
See all →6% HETASTARCH IN 0.9% SODIUM CHLORIDE
6 g/100mL · Injection Solution
HF Acquisition Co LLC, DBA HealthFirst
Available to source
FNG
Liquid
Deseret Biologicals, Inc.
Available to source

Bact Combination
Liquid
Deseret Biologicals, Inc.
Available to source

Bact Combination
Liquid
Deseret Biologicals, Inc.
Available to source
